Section 3 Aquatic Ecology:
Water Properties
- Water is unique in that it is the only natural substance that is found in all three physical states—liquid, solid, and gas—at the temperatures normally found on Earth.
- The solid form, ice, is less dense than the liquid form, which is why ice floats.
- Water is called the "universal solvent" because it dissolves more substances than any other liquid. This means that wherever water goes, either through the ground or through our bodies, it takes along valuable chemicals, minerals, and nutrients.
- The water molecule is highly cohesive—it is very sticky.
- Water has a high specific heat index—it absorbs a lot of heat before it begins to get hot. This is why water is valuable to industries and in your car's radiator as a coolant. The high specific heat index of water also helps regulate the rate at which air changes temperature, which is why the temperature change between seasons is gradual rather than sudden, especially near the oceans.
- Water has a very high surface tension. In other words, water is sticky and elastic, and tends to clump together in drops rather than spread out in a thin film, like rubbing alcohol. Surface tension is responsible for capillary action, which allows water (and its dissolved substances) to move through the roots of plants and through the tiny blood vessels in our bodies.
- The density of water means that sound moves through it long distances (ask a whale!). In sea water at 30°C, sound has a velocity of 1,545 meters per second (about 3,500 miles per hour).
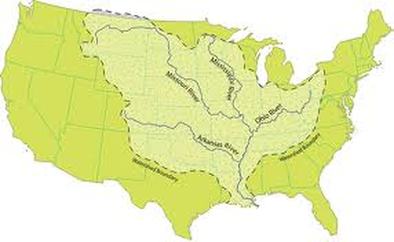
Watersheds: regions on land where water drains into the same region
The Mississippi watershed is...(pause for affect)...Huge!
Ocean Life Zones click below:
http://www.google.com/imgres?imgurl=http://www.geo.arizona.edu/Antevs/nats104/00lect17marinezone.jpg&imgrefurl=http://www.geo.arizona.edu/Antevs/nats104/00lect17marlifzon.html&h=502&w=640&sz=176&tbnid=q2ge5NheDttiCM:&tbnh=115&tbnw=146&zoom=1&usg=__mcVukXrrXZCRmnTXiZtmVxpCdAc=&docid=yWUYg6EM8oiDYM&sa=X&ei=zcydUp7DH83AkQeJsIDwAg&sqi=2&ved=0CC4Q9QEwAA
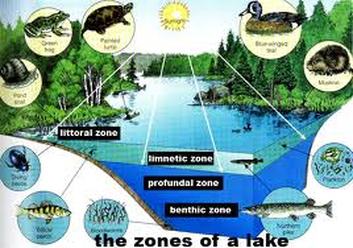
Lake Trophic Levels:
Oligotrophic“Oligo” means very little; therefore, oligotrophic means very little nutrients (Phosphorus and Nitrogen).
The fish found in oligotrophic lakes like cold, high oxygenated water, examples include lake trout and whitefish
In oligotrophic lakes, oxygen is found at high levels throughout the water column. Cold water can hold more dissolved oxygen than warm water, and the deep region of oligotrophic lakes stays very cold. In addition, low algal concentration allows deeper light penetration and less decomposition. Less eutrophication possible.
Mesotrophic“Meso” means middle or mid; therefore, mesotrophic means a medium amount of nutrients (Phosphorus and Nitrogen).
Mesotrophic lakes behave differently than oligotrophic lakes in that they stratify, meaning they separate into layers in the summer. The top layer of water becomes warm from the sun and contains algae. Since the by-product of photosynthesis is oxygen, oxygen concentration remains high at the surface of the lake. The bottom layer remains cooler and can become anoxic in mid-summer.
Eutrophic“Eu” means true; therefore, eutrophic literally means true nutrients or truly nutrient rich (Phosphorus and Nitrogen).
Eutrophic lakes are very fertile from all the nutrients carried into the lake from the surrounding landscape. These nutrients (Phosphorus and Nitrogen) support high densities of algae, fish and other aquatic organisms. Since eutrophic lakes have so much biomass, there is a lot of decomposition occurring at the bottom. This decomposition uses up oxygen, causing the bottom of the lake to become anoxic in the summer.
The fish found in oligotrophic lakes like cold, high oxygenated water, examples include lake trout and whitefish
In oligotrophic lakes, oxygen is found at high levels throughout the water column. Cold water can hold more dissolved oxygen than warm water, and the deep region of oligotrophic lakes stays very cold. In addition, low algal concentration allows deeper light penetration and less decomposition. Less eutrophication possible.
Mesotrophic“Meso” means middle or mid; therefore, mesotrophic means a medium amount of nutrients (Phosphorus and Nitrogen).
Mesotrophic lakes behave differently than oligotrophic lakes in that they stratify, meaning they separate into layers in the summer. The top layer of water becomes warm from the sun and contains algae. Since the by-product of photosynthesis is oxygen, oxygen concentration remains high at the surface of the lake. The bottom layer remains cooler and can become anoxic in mid-summer.
Eutrophic“Eu” means true; therefore, eutrophic literally means true nutrients or truly nutrient rich (Phosphorus and Nitrogen).
Eutrophic lakes are very fertile from all the nutrients carried into the lake from the surrounding landscape. These nutrients (Phosphorus and Nitrogen) support high densities of algae, fish and other aquatic organisms. Since eutrophic lakes have so much biomass, there is a lot of decomposition occurring at the bottom. This decomposition uses up oxygen, causing the bottom of the lake to become anoxic in the summer.
Categories of aquatic life:
WETLANDS:
1. Marshes (wet area with no trees or shrubs)
a. Tidal - some tidal marshes are estuaries, where rivers meet the sea
b. Non-tidal
2. Swamps (wet area with trees or shrubs)
a. forested
b. shrubs
3. Bogs (wet area with acidic peat soil low in nutrients) colder regions
a. fens - bogs fed only from ground water (Fenway Park area!)
Value of Wetlands
http://www.ecy.wa.gov/programs/sea/wetlands/functions.html
All about wetlands http://water.epa.gov/type/wetlands/types_index.cfm
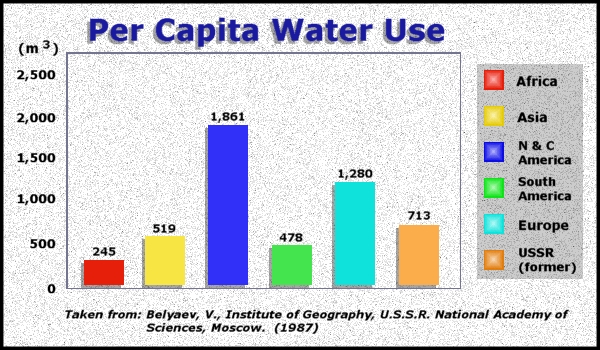
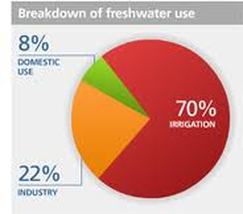
Water Use:
Where do we get water?
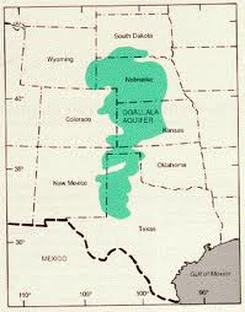
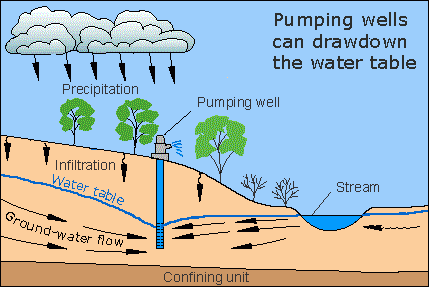
1. Aquifers: Stored groundwater
Ogallala Aquifer: World’s largest aquifer. Holds enough water to cover the U.S. with 1.5 feet of water. Being depleted for agricultural and urban use.
2. Rivers, lakes, streams
Precipitation RECHARGES rivers, lakes, streams and aquifers.
In developed countries water is treated to get rid of sediment and to disinfect with chlorine. It is then sent to homes or stored in large water tanks
3. Desalination (only in wealthy countries)
Aral Sea Disaster:
http://orexca.com/aral_sea.shtml
Groundwater:
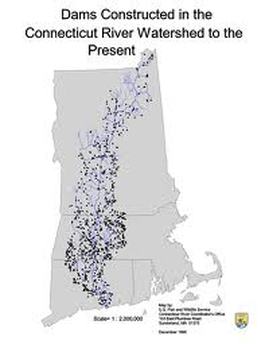
Freshwater has to be diverted with DAMS:
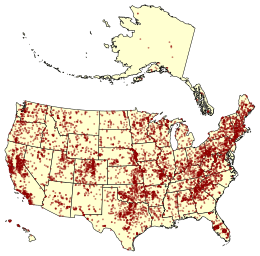
Here are all the dams in the US.
Three Gorges Dam:
http://www.scientificamerican.com/article.cfm?id=chinas-three-gorges-dam-disaster
Are Dams good or bad?
Good:
1. Can give electricity (hydroelectric power like the Hoover or Three gorges in China)
2. Flood control
Bad:
1. You have to move people to flood their land.
2. Stops fish migration.
3. Sediments build-up at the dam site that would normally flow out to sea.
4. Changes the ecosystem downstream because it diverts their water.
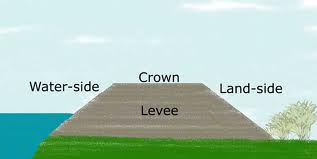
Levees: Allow humans to use land below sea level
Important for agriculture in floodplains and coastal cities like New Orleans
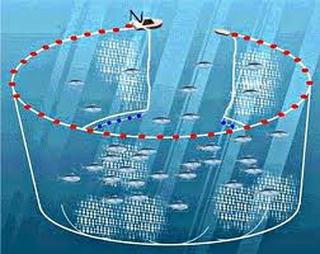
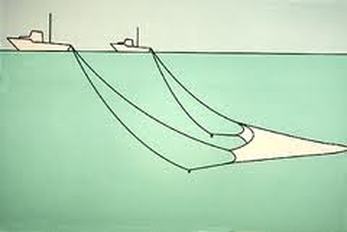
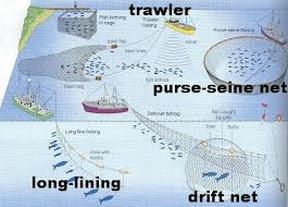
Fishing Practices:
Aquaculture or fish farms:
Overfishing: Tragedy of the commons
http://overfishing.org/pages/Overfishing_in_one_minute.php?w=pages
Water Quality Tests:
Dissolved Oxygen (DO): This test is the most important of the nine water quality tests to measure water's ability to support plants and animals. There are many different factors that affect the amount of dissolved oxygen in water, the main one being temperature. As temperature rises, less gas will dissolve.
Turbidity
Turbidity measures water clarity, which allows sunlight to penetrate to a greater depth. The main sources of turbidity are erosion, living organisms, and those from human endeavors
Total Solids
Total solids measures both dissolved and suspended solids. There are six major types of total solids; silt, clay, soil runoff, plankton, industrial waste, and sewage.
Ph Level
The PH of water is important to aquatic life. If the PH falls below 4 or above 9 everything is dead.
Temperature and Flow Rate
Temperature is a very important part of a river's ecology. There are many natural and human factors that can affect a river's temperature. Human factors include industry, development, and dams. To measure temperature and flow rate you must find two places along the river that are about 1.6 kilometers apart that have the same conditions, then two people measure the temperature at approximately the same time. If the difference is greater than 2 degrees Celsius, then there is thermal pollution. To find flow rate you use a buoyant object, we used an orange, and float it down the river.
Nitrates
Nitrogen is necessary for plant and animal life. Water is tested for nitrates to monitor and control eutrofication , which causes more plant growth and decay.
Fecal Coliform
This nasty stuff is a certain bacteria that propagate in the digestive tracts of humans and animals. They coexist with other bacteria so they are often used as indicators of possible pathogenic contamination. There are many ways fecal coliform can enter a waterway such as animal waste, untreated sewage, combined sewage overflow, and septic tanks.
Biochemical Oxygen Demand
BOD is a measure of oxygen removed from an aquatic environment by aerobic microorganisms. It measures levels of organic pollution in lakes and streams
Phosphorous
Phosphates is a nutrient needed in growth. The phosphate ion is found in shells, bones, and in animal teeth. By removing phosphorous from sewage the amount of phosphate ions in the water will be lowered.
Hard Water:
Water described as “hard” is high in dissolved minerals, specifically calcium and magnesium. Hard water is not a health risk, but a nuisance because of mineral buildup on fixtures and poor soap and/or detergent performance.
Coral Reefs:
How Are Coral Reefs Formed?
Coral reefs are formed by small marine animals (polyps) and plants through the processes of slow accumulation and deposition of calcium carbonate (limestone) extracted from seawater. It is the ultimate symbiotic relationship.
Threats to Coral Reefs:
1. Ocean Acidification
The oceans have absorbed approximately 525 billion tons of carbon dioxide from the atmosphere, or about one third of the anthropogenic carbon emissions released. The pH of ocean surface waters has already decreased by about 0.1 units from an average of about 8.21 to 8.10 since the beginning of the Industrial Revolution.
When CO2 reacts with seawater it forms carbonic acid
2. Coral Bleaching
The increase in water temperature causes coral polyps, stressed by heat, to expel the algae that live within them. These algae, called zooxanthellae (zo-zan-THEL-ee) normally provide the coral with up to 80% of its energy, making zooxanthellae essential for coral survival. The algae are also normally responsible for the color of coral, so when they are expelled, the coral appears white or "bleached."
3. Water Pollution
Oil, gas and pesticide contamination
4. Sedimentation blocks
Construction along coasts, inshore construction, mining, logging and farming along coastal rivers can all lead to erosion. As a result, particles end up in the ocean and cover coral reefs. This 'smothers' coral and deprives it of the light it needs to survive. Mangrove trees and seagrasses, which normally act as filters for sediment, are also being rapidly destroyed. This has led to an increase in the amount of sediment reaching coral reefs. Mangrove forests are often cut for firewood or removed to create open beaches. They are also destroyed by prawn harvesters to open up areas to create artificial prawn farms.
5. Coastal Development
6. Overfishing
Top predators are taken and have an impact on the whole ecosystem
7. Careless Tourism
8. Ozone Depletion
The destruction of the ozone layer is caused by the presence of chlorofluorocarbons (CFCs). This presence causes the depletion of protective ozone in the atmosphere and increases the intensity and nature of ultraviolet radiation that reaches the earth's surface. Although corals have a natural sunscreen to protect themselves from the tropical sun, most scientists believe that increased levels of ultraviolet radiation damage coral in shallow areas.
How Are Coral Reefs Formed?
Coral reefs are formed by small marine animals (polyps) and plants through the processes of slow accumulation and deposition of calcium carbonate (limestone) extracted from seawater. It is the ultimate symbiotic relationship.
Threats to Coral Reefs:
1. Ocean Acidification
The oceans have absorbed approximately 525 billion tons of carbon dioxide from the atmosphere, or about one third of the anthropogenic carbon emissions released. The pH of ocean surface waters has already decreased by about 0.1 units from an average of about 8.21 to 8.10 since the beginning of the Industrial Revolution.
When CO2 reacts with seawater it forms carbonic acid
2. Coral Bleaching
The increase in water temperature causes coral polyps, stressed by heat, to expel the algae that live within them. These algae, called zooxanthellae (zo-zan-THEL-ee) normally provide the coral with up to 80% of its energy, making zooxanthellae essential for coral survival. The algae are also normally responsible for the color of coral, so when they are expelled, the coral appears white or "bleached."
3. Water Pollution
Oil, gas and pesticide contamination
4. Sedimentation blocks
Construction along coasts, inshore construction, mining, logging and farming along coastal rivers can all lead to erosion. As a result, particles end up in the ocean and cover coral reefs. This 'smothers' coral and deprives it of the light it needs to survive. Mangrove trees and seagrasses, which normally act as filters for sediment, are also being rapidly destroyed. This has led to an increase in the amount of sediment reaching coral reefs. Mangrove forests are often cut for firewood or removed to create open beaches. They are also destroyed by prawn harvesters to open up areas to create artificial prawn farms.
5. Coastal Development
6. Overfishing
Top predators are taken and have an impact on the whole ecosystem
7. Careless Tourism
8. Ozone Depletion
The destruction of the ozone layer is caused by the presence of chlorofluorocarbons (CFCs). This presence causes the depletion of protective ozone in the atmosphere and increases the intensity and nature of ultraviolet radiation that reaches the earth's surface. Although corals have a natural sunscreen to protect themselves from the tropical sun, most scientists believe that increased levels of ultraviolet radiation damage coral in shallow areas.
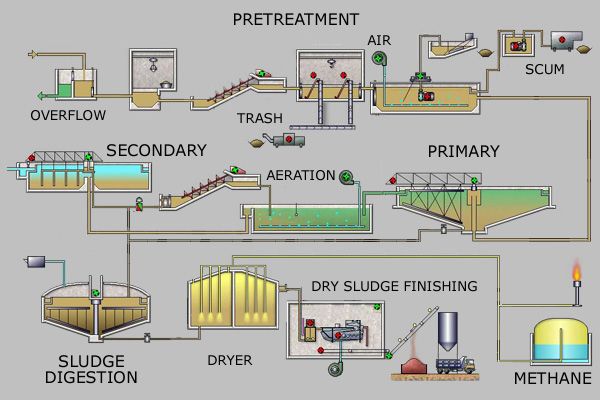
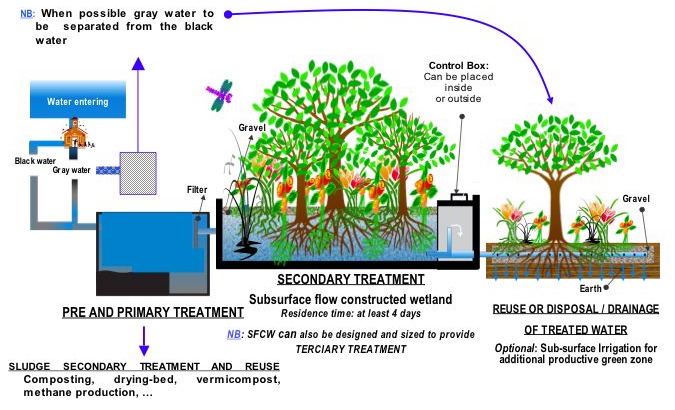
Sewage treatment generally involves three stages, called primary, secondary and tertiary treatment.
- Primary treatment (Mechanical treatment) bars and screens grab garbage. What's left is split into three parts. Solids settle to the bottom and become SLUDGE; while oil, grease and lighter solids float to the surface and become SCUM. The liquid is passed on...
- Secondary treatment (biological treatment) air is added to the liquid to supply to the microorganisms so they can digest the waste. The sludge becomes fertilizer. (Boston actually sells its sludge to Tropicana, the orange juice people)
- Tertiary treatment what's left is chlorinated or UV rayed to kill microorganism and then it is let back into the environment
Types of Water Pollution
Water pollution can affect groundwater (aquifers etc.) and surface water (oceans, lakes, rivers, streams, etc)
What's polluting water?: Farm pesticides and fertilizers;
industrial chemicals (making paints, plastics, leathers, etc);
power plant effluent (hot water); power plant gases returning as acid rain;
sewage; old coal and metal mines (AMD); landfill seepage; oil spills;
1. Nutrient Pollution
Nutrients are usually found in sewage and fertilizers.
These can cause excess vegetation growth in the water of algae and weeds and increase the BOD. By increasing the BOD, the DO levels come way down causing "fish-kills" and Cultural EUTROPHICATION. Eutrophication is enhanced when organic material is decomposed using the remaining oxygen.
(hypoxia - low oxygen; anoxia - no oxygen)
Acid mine drainage (AMD) is when old and abandoned mines supply surface runoff with nutrient and chemical pollutants. Some gets into the groundwater.
2. Chemical Pollution
A. Organic (chemicals containing Carbon): solvents, plastics, landfill seepage
B. Inorganic (chemicals not containing Carbon): Heavy metals
Toxins build up as they move up the food chain BIOACCUMULATION. They then effect organisms BIOMAGNIFICATION
Sharks and top carnivores sometimes can be considered hazardous waste.
3. Thermal Pollution
Water from a power plant (coal or nuclear) is heated and dumped into river systems
4. Microbiological Pollution
This is a significant problem for people in third world countries without sewage treatment who have no clean drinking water and/or facilities to purify the water. Cholera etc.
5. Sediment pollution: Over-grazed range land, strip mines, over logging, etc. causes sediments to runoff into water bodies
Two Sources of Water pollution:
I. Point-source - meaning it comes from a specific place in space and time you can ID (tailpipe; smokestack)
II. Non-point source - general pollutant (agricultural runoff; CO2 build-up)
Exxon Valdez
http://www.eoearth.org/article/Exxon_Valdez_oil_spill
Water Pollution Legislation